HSP Basics
Like, as we all know, attracts like. So if we have a formula for "like" we can judge the solubility of a polymer in a solvent or the compatibility of a polymer and plasticiser or the stability of a nanoparticle in a solvent dispersion by how alike they are.
It starts with evaporation
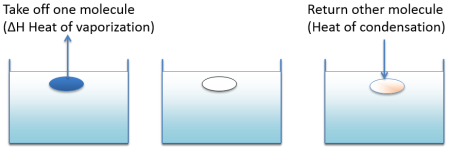 It takes energy (enthalpy of vapourisation) to evaporate a solvent. You get all that energy back when you condense it. If you evaporate a portion of a solvent and then condense in an equal volume of a different solvent, if the two solvents are alike then the energy cost of doing this is minimal and the solvents are miscible. If they are very different then energy in and out don't balance and the miscibility is low.
It takes energy (enthalpy of vapourisation) to evaporate a solvent. You get all that energy back when you condense it. If you evaporate a portion of a solvent and then condense in an equal volume of a different solvent, if the two solvents are alike then the energy cost of doing this is minimal and the solvents are miscible. If they are very different then energy in and out don't balance and the miscibility is low.
It's more precise to talk of the cohesive energy, E, of a solvent, i.e. how much energy is required to break it apart into gas molecules. If E is broken into 3 components of Dispersion, Polar and Hydrogen-bonding then we get:
E = ED + EP + EH
We are generally not familiar with cohesive energy. Instead we know the latent heat of vaporization Hv, R the universal gas constant, and T the absolute temperature . The relationship between them is:
E = HV - RT
Cohesive energy is not so useful on its own. Bigger molecules in a molecular series, not surprisingly, have bigger cohesive energies. To scale them so that molecules in a series are more comparable we need the cohesive energy density which is obtained by dividing by molar volume V:
E/V = ED/V + EP/V + EH/V
The cohesive energy density is more conveniently handled in terms of the solubility parameter δ where δ²=E/V. This gives us the classic formula for Hansen Solubility Parameters where the total parameter, δ, is broken into δD, δP and δH for Dispersion, Polar and Hydrogen-bonding:
δ² = δD² + δP² + δH²
The units of solubility parameters are MPa½. Older references use (cal/cc)½ which are a factor of 2.0455 smaller.
The measure of "like"
The HSP Distance between two molecules, conventionally called Ra, is the measure of how alike they are. The smaller Ra, the more likely they are to be compatible. The famous formula, used for nearly 50 years is:
Ra² = 4(δD1-δD2)² + (δP1-δP2)² + (δH1-δH2)²
The factor of 4 in front of the ΔδD term has aroused controversy for decades but plausible theoretical reasons and overwhelming experimental evidence show that it is valid. Conveniently it makes HSP plots into nice spheres. If from measurements of "good" and "bad" solvents you can define a sphere of radius Ro which contains all the good solvents, then you can define a Relative Energy Difference, RED, which tells you where you are in solubility space:
RED = Ra/R0
Hansen Solubility Parameters in Practice
If you know the HSP of your solvents, polymers, plasticisers, pigments, nanoparticles, bio-system... then it is very easy to use the Distance formula to understand solubility, swellability, dispersability, permeation and so forth. You can find plenty of published HSP values on the internet. If there is no solvent with the right balance of HSP, cost, safety, odour etc. (and these days it is very hard to find such solvents) you can create your own by mixing solvents, using the rule that the HSP of the mix is the volume-weighted average of the HSP of the components. And Hansen's method of measuring HSP of unknowns via simple good/bad tests with a range of solvents and fitting to a Sphere is not hard to do. The Excel spreadsheet, improved in 2019 by Dr Manuel Díaz de los Ríos, Director of Derivatives Division, ICIDCA (acknowledged with many thanks!) HSP_Calculations.xlsx allows you to try out many of the basics.
There is a full, Open Source, paper on using the spreadsheet technique you can download from here, Manuel Díaz de los Ríos, Eduardo Hernández Ramos, Determination of the Hansen solubility parameters and the Hansen sphere radius with the aid of the solver add‑in of Microsoft Excel, SN Applied Sciences (2020) 2:676
The famous Pirika site from Dr Hiroshi Yamamoto provides lots of insights into HSP along with a number of handy apps for carrying out calculations
However, many of the published values are suspect as they use different methods (van Krevelen, Hoy etc.) to estimate HSP values. And in reality it is hard to assemble all the information you need to solve your specific problem. That's why so many people are using HSPiP with its huge, consistent datasets and its powerful range of calculation/optimisation tools.